|
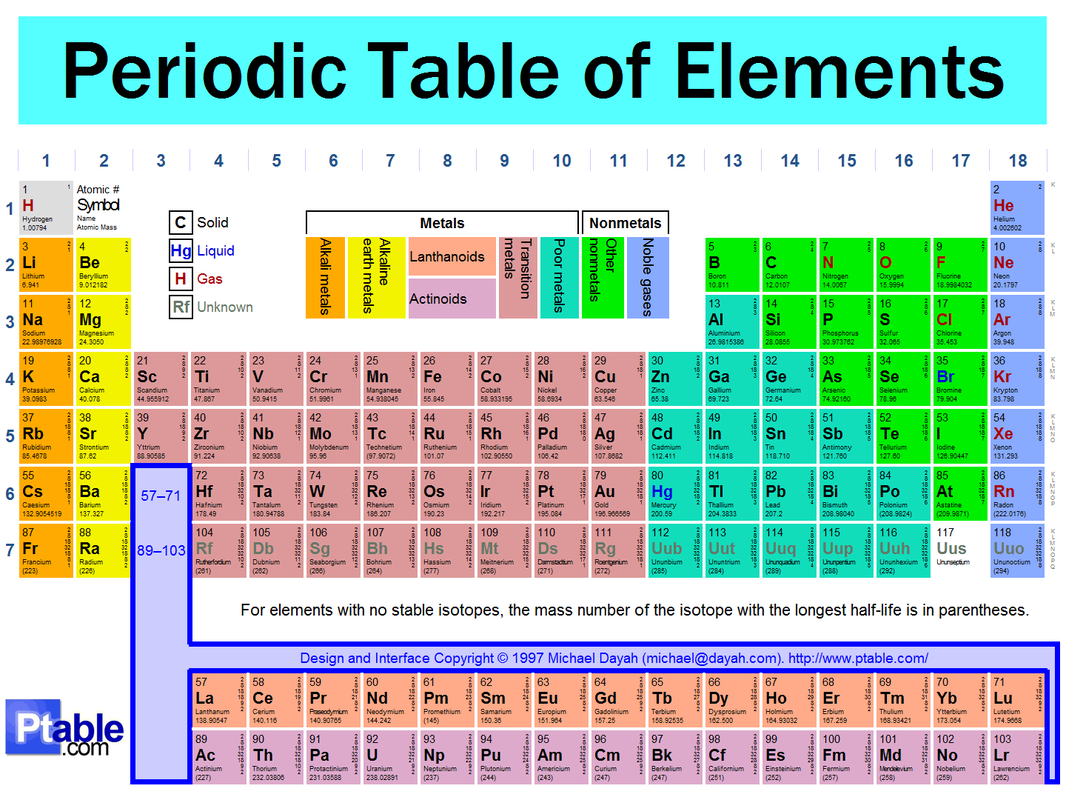
Periods are the rows of elements in the period table. They tell you how many shells, or energy levels, an element has. For example: period 1 has one shell, period two has two shells, period three has three shell, etc.
|
Elements in the periodic table are mostly arranged by atomic number, valence electrons, and energy levels of elements. The atomic number (the large number above the atomic symbol) is the number of protons an atom has. You'll notice that the periodic table goes in order of atomic number. The reason the periodic table looks so weird is because its divided by valence electron (groups) as well as by energy level (periods). The periodic table has three types of elements: metal, nonmetal, and metalloids. The metals are the majority of the table and are located on the left. The nonmetals are all in the upper right corner of the table except for Hydrogen which in the upper left corner. The Metalloids are located somewhat in between the metals and nonmetals. The atomic mass of an element is located under the name of the element and can tell us how many neutrons are in an element by subtracting the atomic number from the atomic number.
|
Groups are the columns on a periodic table. They tell you how many valence electrons an element has except for columns 3-12. For example: column 1 has one valence electron, column two has two, column thirteen has three, column four has four, etc.
|